Skeaff S. and Gibson, R.S.,
Nutritional Assessment,
Iodine
3rd Edition April, 2026
Abstract
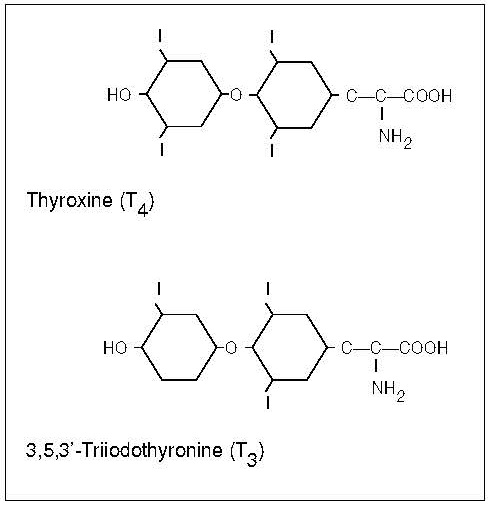
Iodine is an essential trace element required for the synthesis of the thyroid hormones thyroxine and triiodothyronine, which are needed for normal growth, neurodevelopment, and many important metabolic processes throughout life. Assessment of iodine status should be included in nutritional surveillance programmes and as an important component of public health policy, particularly in places where iodine deficiency has been observed. This chapter provides a comprehensive overview of the principles, methods, and challenges involved in assessing iodine nutrition, with a focus on biochemical, functional, and clinical indicators. Urinary iodine concentration (UIC), derived from spot or 24 hour urine samples, remains the most widely used biomarker for evaluating recent iodine intake in populations, reflecting the close relationship between dietary iodine intake and urinary excretion. Strengths and limitations of UIC are discussed, including its high intra individual variability and its unsuitability for diagnosing iodine status in individuals. Other indicators of iodine status, including thyroid volume assessed by palpation or ultrasonography, neonatal thyroid stimulating hormone concentrations, and thyroglobulin, are evaluated, highlighting their use across different settings, age groups, and severity of iodine deficiency. Thyroglobulin and breast milk iodine concentration hold promise as complementary biomarkers, particularly in the context of monitoring mild deficiency and iodine status during pregnancy and lactation. The chapter emphasises the need for careful selection and combined use of indicators, quality assured laboratory methods, and appropriate interpretive criteria as recommended by the World Health Organisation and Iodine Global Network. Ongoing surveillance of iodine status is essential, as populations remain vulnerable to both iodine deficiency and excess despite successful salt iodisation programmes. CITE AS: Skeaff S. and Gibson. R. S. Principles of Nutritional Assessment: Iodine https://nutritionalassessment.org/iodine/Email: sheila.skeaff@otago.ac.nz
Licensed under CC-BY-4.0
( PDF ) ( epub )